What Is In Your Makeup? Lead Chemicals
The Toxic Twelve Chemicals and Contaminants in Cosmetics
The Toxic Twelve Chemicals and Contaminants in Cosmetics
By Scott Faber, Senior Vice President of Government Diplomacy
Updated Tuesday, May v, 2020
Chemicals and contaminants linked to cancer can exist found in nutrient, h2o and many other everyday products. Still, no category of consumer products is subject field to less government oversight than cosmetics and other personal care products. Although many of the chemicals and contaminants in cosmetics and personal care products likely pose little risk, exposure to some has been linked to serious health bug, including cancer.
Since 2009, 595 cosmetics manufacturers have reported using 88 chemicals, in more than than 73,000 products, that take been linked to cancer, birth defects or reproductive harm.[one]
Many of these chemicals should be banned from cosmetics, every bit proposed in California Assembly Bill 2762, the Toxic-Free Cosmetics Human action. Among the toxic chemicals[2]that should be banned are:
- Formaldehyde, a known carcinogen.
- Paraformaldehyde, a type of formaldehyde.
- Methylene glycol, a blazon of formaldehyde.
- Quaternium xv, which releases formaldehyde.
- Mercury, which can harm the kidneys and nervous organization.
- Dibutyl and diethylhexyl phthalates, which disrupt hormones and damage the reproductive organisation.
- Isobutyl and isopropyl parabens, which disrupt hormones and harm the reproductive organisation.
- The long-chain per- and polyfluoroalkyl substances known every bit PFAS, which accept been linked to cancer.
- M- and o-phenylenediamine, used in hair dyes, which irritate and sensitize the peel, damage Dna and can cause cancer.
All of these toxic chemicals have been banned past the Eu and many other nations, and many have been slated for removal from the store brands of major U.S. retailers, including Target, Rite Aid, Walgreens and CVS Health.[3] For example, equally of the end of 2019, CVS Wellness prohibits the employ of formaldehyde, many chemicals that release formaldehyde, many parabens, dibutyl phthalate and diethylhexyl phthalate. Some of these are already banned from products sold in Whole Foods.[4]
Some of the chemicals included in A.B. 2762 do not accept to exist disclosed on the package, because they are components of fragrance and so are exempt from federal labeling requirements. In particular, two phthalates included in A.B. 2762 – dibutyl phthalate and diethylhexyl phthalate – may exist included in combinations of chemicals disclosed on the label every bit fragrance but do not have to be disclosed as private chemicals.
Dibutyl phthalate is an endocrine disruptor and a developmental toxicant that harms male person reproductive organization development. Information technology can crusade early on puberty in boys and other changes in the reproductive system. Diethylhexyl phthalate harms the reproductive system and can affect the developing fetus. It has as well been classified by the International Bureau for Research on Cancer as perhaps carcinogenic.[five]
Many of these chemicals are rarely used in personal care products and volition be like shooting fish in a barrel to replace. For example, in Pare Deep®, EWG'south database of cosmetics and other personal care products, EWG found isobutylparaben and isopropylparaben in just 96 and 12 products, respectively, offered for sale since January 2017. A 2018 analysis past EWG found fewer than 200 products contained ane of 13 PFAS chemicals.[6]
The cosmetics industry has grown dramatically since 1938, when Congress last enacted cosmetics legislation, the Food, Drug, and Cosmetic Act. At that fourth dimension, the cosmetics industry reported approximately $one billion in sales.[7] In 2016, it reported more than $169 billion in sales.[8] Nevertheless, but ii pages of the 829-folio human action governs cosmetics, and those provisions provide the Food and Drug Administration no financial resources and sharply limit its authority to regulate chemicals and contaminants that pose chronic risks.[ix]
Although Congress has since then given the FDA the power to ensure that food additives,[10] color additives[11]and pesticides[12] pose "no harm" from repeat exposures, Congress has not given the agency the same authority to regulate the chronic risks posed by chemicals and contaminants in cosmetics. Instead, the FDA largely relies on the personal care products industry to regulate itself equally a style to address the risks its products pose.
Consumers use a wide variety of cosmetics and personal care products. Few consumer products contribute as many chemical exposures as cosmetics and other personal intendance products. Each day, American women use an average of 12 personal care products that contain 168 different chemicals. Men utilise an average of six personal care products that contain 85 different chemicals.[thirteen]Many of these products are applied straight to the skin, the torso'due south largest organ, where ingredients can be captivated directly into the bloodstream.[14]
Consumer apply of cosmetics continues to grow dramatically. Since 2010, the U.S. cosmetics market has grown an average of 4.1 percent annually, and internet sales take expanded to business relationship for 8.4 percentage of the total marketplace share.[15]
Cosmetic imports are also on the rise. In fiscal year 2016, 2.9 one thousand thousand lines of cosmetics were imported into the U.S. from 181 dissimilar countries.[sixteen] Lines of corrective imports doubled over the past decade, with a substantial increase after FY 2011.[17]Corrective imports from Cathay increased past 79 percent between FY 2011 and FY 2016.
Although almost chemicals in cosmetics pose little or no run a risk, some take been linked to serious health issues, including cancer, reproductive and neurological harm, and developmental delays. Corrective chemicals enter the body through the pare, inhalation, ingestion and internal use, and pose the same risks equally nutrient chemicals.[18] In improver to the risks posed by intentionally added ingredients, cosmetics tin exist contaminated with heavy metals, including arsenic, cadmium, pb, mercury and nickel.
Some chemicals used in personal care products pose risks at very low doses[22]and tin interfere with the hormone system.[23] Research shows that "endocrine disrupting" chemicals such as parabens and phthalates may pose the greatest risk during prenatal and early postnatal development, when organ and neural systems form.[24] Exposure to these chemicals has been linked to endocrine diseases and some types of cancer.[25] For case, endocrine disruptors are known to touch how women's bodies use estrogen and thus have been linked to chest cancer.[26] Research has also shown that endocrine disruptors can harm the allowed organization – an effect that makes the states more susceptible to illness and viruses.
Many cosmetics have likewise been linked to acute risks, including burns and infections. Formaldehyde-based hair-straightening procedures, referred to every bit "keratin treatments," take been linked to hair loss, rashes, blisters, nosebleeds, bleeding gums and loss of taste and smell.[27]Other cosmetics accept caused hair loss.[28]
The FDA continues to find cosmetics contaminated with bacteria, including body wash, face powders, shadows and lotions, or containing banned colors chemicals, including shampoos, soaps, cleaners and temporary tattoos.[29]
Under the current police force,the FDA has little authorization to review chemicals in cosmetics and other personal intendance products.Personal care products companies do not take to register with the FDA, provide the FDA with ingredient statements, adopt Skillful Manufacturing Practices, or GMPs, report adverse events to the FDA, or provide the FDA with access to prophylactic records.
The FDA does not have the power to suspend registration or order recalls when products pose a gamble of serious adverse health consequences or death.By contrast, manufacturers of food, drugs and medical devices must register with the FDA, maintain and give the FDA access to records and report adverse events. If food, drugs or devices are unsafe, the FDA can suspend production and production licenses. If unsafe food or devices reach the marketplace, the FDA can club a recall and take legal action confronting drugmakers that exercise not call back their products.[30]
Of more 10,000 chemicals used to formulate cosmetics, just xi have ever been banned or restricted by the federal Nutrient and Drug Administration. By contrast, the FDA, the Environmental Protection Bureau and the Consumer Product Condom Committee have broad authority to ensure the safety of chemicals in other consumer products.
For case, the FDA has the authority to review chemicals in prescription[31] and over-the-counter drugs,[32] and chemicals institute in food,[33] and the EPA has the dominance to review chemicals in pesticides used in homes and on farms[34] and to set limits for pesticide residues on food.[35] In 2016, Congress expanded EPA authority to review chemicals in cleaners, paints, solvents and many other consumer products.[36]
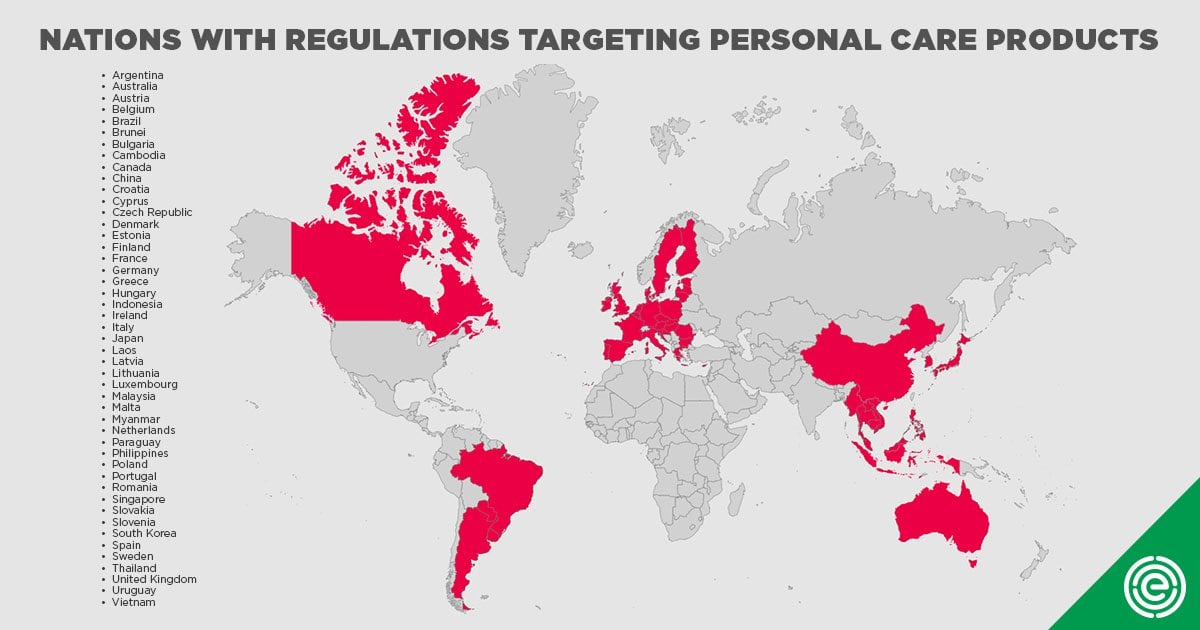
The U.S. has also fallen far behind our international trading partners in the regulation of cosmetics. More than 40 nations have taken steps to ban or restrict, in combination, more 1,400 chemicals or contaminants in cosmetics and personal care products, including chemicals linked to cancer, reproductive impairment and neurological harm.[37]
All of the chemicals identified in A.B. 2762 take already been banned by the EU, including isopropylparaben and isobutylparaben, dibutyl phthalate and diethylhexyl phthalate,[38] mercury,[39] formaldehyde and many PFAS. Other nations have restricted the presence of chemicals such equally formaldehyde[twoscore] and perfluorooctanoic acid (PFOA).[41]
The cosmetics industry has long fought meaningful oversight. Since the early 1950s, it has defeated efforts by Congress to modernize cosmetics law.[42] Since 2015, some cosmetics companies have supported giving the FDA the authority and resources to review and regulate chemicals and contaminants of concern in cosmetics, and take supported requiring manufacturers to annals, provide ingredient statements, adopt GMPs and report adverse events.
Some companies accept likewise supported giving FDA the power to suspend product of dangerous products and order mandatory recalls.[43] However, other companies accept not supported FDA review and oversight.
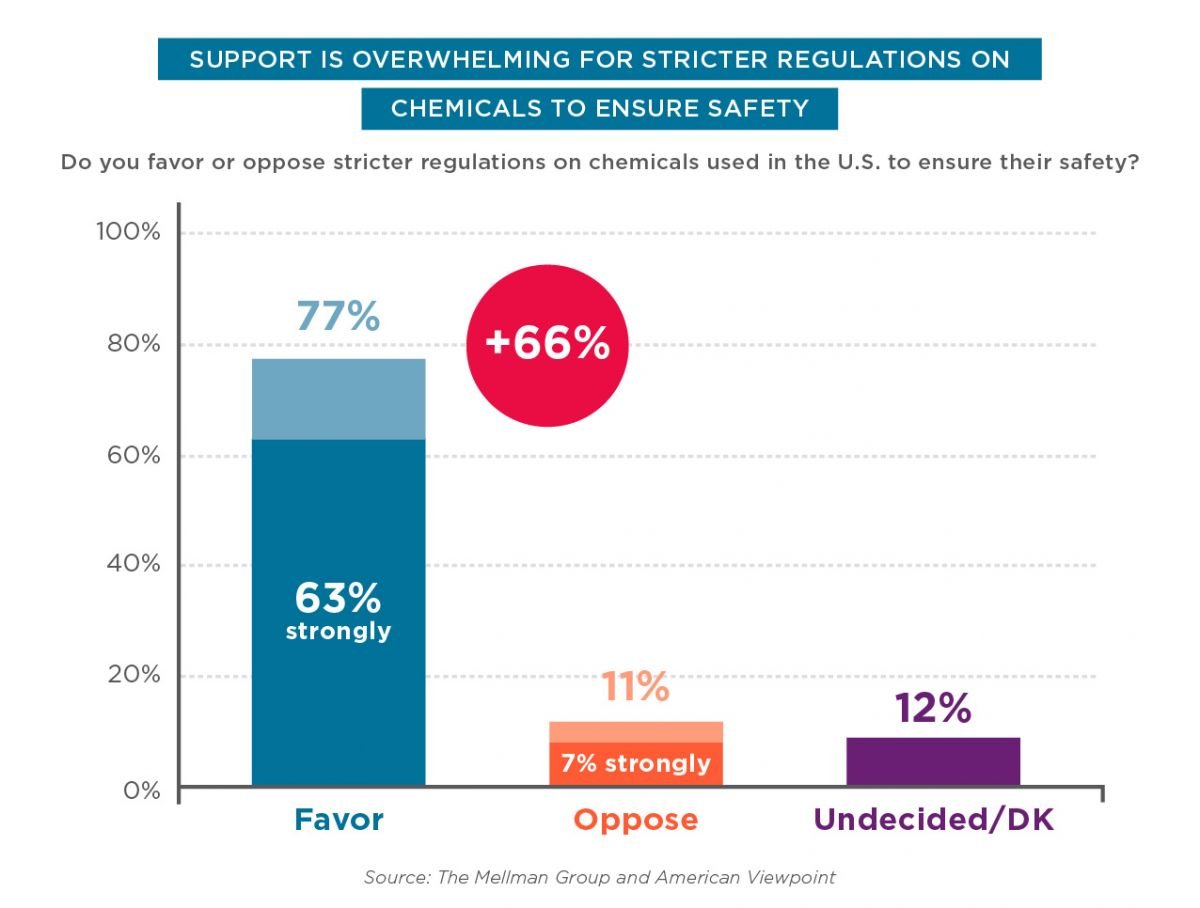
Consumers overwhelmingly back up federal oversight of cosmetic chemicals. Two-thirds of consumers believe chemicals in cosmetics are already reviewed by the FDA, according to polling conducted past American Viewpoint and the Mellman Group in 2016.[44]Iii-fourths of consumers – regardless of historic period, race or party amalgamation – support stricter oversight of chemicals in cosmetics, and nearly nine in 10 consider stricter rules very of import.
In addition, nine in ten consumers believe cosmetic companies should accept to notify the FDA if their products harm consumers; back up giving the FDA mandatory retrieve authorisation; and support rules ensuring cosmetics are produced in clean environments.
[i] Cal. Dep't of Pub. Health, Cal. Safe Cosmetics Program, Current Data Summary, https://www.cdph.ca.gov/Programs/CCDPHP/DEODC/OHB/CSCP/Pages/SummaryData.aspx (last accessed Mar. 8, 2019). The California Rubber Cosmetics Deed of 2005 requires corrective manufacturers to disclose to the California Department of Public Wellness all products containing ingredients known or suspected to crusade cancer, nativity defects or other reproductive toxicity every bit determined past sure authoritative scientific bodies, including the Ecology Protection Agency, the National Toxicology Program and the International Bureau for Inquiry on Cancer.
[two] Ecology Working Group, the Toxic Xx Cosmetic Ingredients and Contaminants, accessed at http://cdn3.ewg.org/sites/default/files/u352/Toxic%2020%20List.pdf?_ga=ii.36293026.364182527.1554126027-937396664.1520601435&_gac=one.259664632.1553597903.CjwKCAjwm-fkBRBBEiwA966fZF139RiUrtNyeYyElYAnOqpk3GtURCnrAjqAsTCXwDcJ1oSIgOtY1BoCOKcQAvD_BwE
>[three] Target, Chemicals, https://corporate.target.com/corporate-responsibility/planet/chemicals (concluding visited Apr. 2, 2019); Rite Aid, Chemical Policy: Restricted Substances List, https://world wide web.riteaid.com/documents/45609410/184110402/rite-aid-restricted-substances-list_1809.pdf/39450b19-0512-73c1-3afb-358987ea5ff6 (last visited Apr. 2, 2019); Walgreens Boots Alliance, Restricted Substances List, https://www.walgreens.com/images/adaptive/si/pdf/WalgreensBootsAllianceRestrictedSubstancesList_11082018.pdf (last visited Apr. ii, 2019); CVS Health, Store Brand Restricted Substances List, https://cvshealth.com/sites/default/files/cvs-health-restricted-chemical-list-by-category.pdf (last visited April. 2, 2019).
[four] Whole Foods, Our Body Intendance Quality Standards, https://www.wholefoodsmarket.com/virtually-our-products/body-care-quality-standards (final visited Apr. 2, 2019); Whole Foods, Premium Body Care Unacceptable Ingredients, https://world wide web.wholefoodsmarket.com/premium-body-care-unacceptable-ingredients (final visited Apr. 2, 2019).
[5] Poursafa, P., Ataei, E., & Kelishadi, R. (2015). A systematic review on the effects of environmental exposure to some organohalogens and phthalates on early puberty.J Res Med Sci, xx(6): 613-18, https://world wide web.ncbi.nlm.nih.gov/pmc/articles/PMC4621657/; Mylchreest, E., Cattley, R.C., & Foster, P.Yard. (1998). Male reproductive tract malformations in rats following gestational and lactational exposure to Di(due north-butyl) phthalate: an antiandrogenic mechanism?Toxicol Sci, 43(1): 47-60, https://world wide web.ncbi.nlm.nih.gov/pubmed/9629619; Kay, V.R., Flower, M.S., & Foster. W.Chiliad. (2014). Reproductive and developmental effects of phthalate diesters in males.Crit Rev Toxicol, 44(6): 467-98, https://world wide web.ncbi.nlm.nih.gov/pubmed/24903855; Int'l Agency for Research on Cancer, Di(2-ethylhexyl) Phthalate (2013), https://monographs.iarc.fr/wp-content/uploads/2018/06/mono101-006.pdf.
>[6] David Andrews and Carla Burns,Is Teflon in Your Cosmetics? Envtl. Working Grp. (Mar. xiv, 2018), https://world wide web.ewg.org/skindeep/2018/03/07/is-teflon-in-your-cosmetics/.
[7] Kerry A. Harnett,Appearing Modern: Women's Bodies, Dazzler, and Ability in 1920s America(Apr. 2009), https://dlib.bc.edu/islandora/object/bc-ir:102409/datastream/PDF/view.
[8] Personal Care Products Council,Economic and Social Contributions Report (2016), https://www.personalcarecouncil.org/wp-content/uploads/2018/x/PCPC_Economic_Social_Contributions_Report_Web.pdf;see besides Statista,Revenue of the Cosmetics Manufacture in the United States from 2002-2016, https://www.statista.com/statistics/717673/cosmetics-personal-care-products-markets-revenue/https://www.statista.com/statistics/243742/revenue-of-the-cosmetic-industry-in-the-usa/ (concluding visited Mar. 2019).
[9]Section 601(a) of the FDCA (21 U.s.a.C. § 361(a)) states that a cosmetic is deemed adulterated if information technology "bears or contains any poisonous or deleterious substance which may render it injurious to users under the conditions of use prescribed in the labeling thereof, or under such conditions of use as are customary or usual." A cosmetic is also adulterated under the 1938 FDCA if packed in unsanitary conditions that may render it "injurious to wellness" or its container is composed in whole or part of whatsoever poisonous or deleterious substance that may return information technology "injurious to wellness."
[10] Food Additives Amendment of 1958, Pub. L. No. 85-929, 72 Stat. 1784.
[11] Color Additives Amendment of 1960, Pub. L. No. 86-618, 74 Stat. 397.
[12] Food Quality Protection Act of 1996, Pub. L. No. 104-170, 110 Stat. 1489.
[xiii] Envtl. Working Grp.,Exposures Add Upwardly- Survey Results (2004), http://world wide web.ewg.org/skindeep/2004/06/15/exposures-add together-up-survey-results/.
[fourteen] Marcy Laub,Skin deep,Harvard Univ. (2015), https://green.harvard.edu/news/peel-deep.
[xv]Nilesh Rajput,Cosmetics Market by Category,Global Opportunity Analysis and Manufacture Forecast, 2014 - 2022,Centrolineal Market place Research (July 2016),https://world wide web.alliedmarketresearch.com/cosmetics-market;run into also Statista,Cosmetics Industry – Statistics & Facts (2016), https://www.statista.com/statistics/297070/growth-rate-of-the-global-cosmetics-market/; Euromonitor Intl.,Colour Cosmetics in the US (May 2017) http://www.euromonitor.com/colour-cosmetics-in-the-united states of america/written report.
[16]Come across Letter of the alphabet from Anna G. Abram, Deputy Comm'r for Policy, Planning, Legislation and Analysis, Food & Drug Admin., to Representative Frank Pallone, Ranking Member, House Energy and Commerce Comm. (June 30, 2017), https://democrats-energycommerce.house.gov/sites/democrats.energycommerce.house.gov/files/documents/FDA%20Response%20to%20Rep.%20Pallone%20on%20Cosmetic%20Imports.pdf.
>[17]Id.
[18] Encounter mostly UNL Envtl. Health and Safety,Toxicology And Exposure Guidelines(Jan. 2003), https://ehs.unl.edu/documents/tox_exposure_guidelines.pdf.
[19] Geologically, talc and asbestos can exist formed from the aforementioned parent rock. As a result, mined talc deposits in many parts of the world can exist contaminated with asbestos fibers.
[20] Cosmetics companies are non required to register and provide ingredient statements to FDA, so FDA is unable to estimate the number products that incorporate talc.
[21] Envtl. Working Grp., EWG's Peel Deep® Cosmetics Database, https://www.ewg.org/skindeep/.
<
[22] Laura North. Vandenberget al., Hormones & Endocrine-Disrupting Chemicals: Low-Dose Furnishings & Nonmonotonic Dose Responses, 33 Endocrine Rev. 378-455 (2012), http://www.ncbi.nlm.nih.gov/pubmed/22419778.
[23] Chemicals like phthalates and triphenyl phosphate tin can disrupt the hormone organization past mimicking or blocking a natural hormone. When an endocrine-disrupting chemical mimics a hormone, the chemic tricks the hormone'south receptor into thinking the chemical is the hormone. When the chemical blocks a hormone, the chemical can bind to a receptor and the hormone may not be activated. Run intoVeldhoen Due northet al.,The bactericidal agent triclosan modulates thyroid hormone-associated gene expression and disrupts postembryonic anuran development,Aquat Toxicol (June 2007),https://www.ncbi.nlm.nih.gov/pubmed/17011055.
[24] See, east.m.,Nat'l Inst. of Envtl. Health Sci.,Endocrine Disruptors, https://world wide web.niehs.nih.gov/wellness/topics/agents/endocrine/index.cfm (last visited Apr. 2, 2019).
[25]E.g., Andrea C. Goreet al.,Executive Summary to EDC-2: The Endocrine Society'due south Second Scientific Statement on Endocrine-Disrupting Chemicals, 36 Endocrine Rev. 593-602 (2015), http://press.endocrine.org/doi/10.1210/er.2015-1093.
[26] Id.
[27]Encounter,e.g., Envtl. Working Grp.,Brazilian-Fashion Blowouts: Nevertheless Poisonous, Still in Salons (2015), http://www.ewg.org/enviroblog/2015/08/brazilian-manner-blowouts-withal-poisonous-yet-salons.
[28] Run into, eastward.yard.,Eric Lipton & Rachel Abrams,Their Hair Barbarous Out. Should the F.D.A. Accept the Power to Human action?Northward.Y. Times, (Aug. 2016), http://www.nytimes.com/2016/08/16/us/politics/cosmetics-industry-congress-regulation-wen.html?_r=0.
[29] Scott Faber,Contaminated Cosmetics Pose Growing Take chances to Consumers, Envtl. Working Grp. (Feb. 2018), https://world wide web.ewg.org/news-and-assay/2018/02/contaminated-cosmetics-pose-growing-risk-consumers#.Wp65FpPwZ-V.
[30]Due east.chiliad., 21 U.s.C. § 350(d) (nutrient); 21 C.F.R. § 807 (devices); 21 United states of americaC. § 360 (drugs); 21 C.F.R. §§ 607.65, 1271 (biologics).
[31] 21 U.Southward.C. §§ 351-360fff.
[32] 21 C.F.R. § 330.
[33] 21 United states of americaC. § 348.
[34] vii The statesC. §§ 136a-136d.
[35] 21 U.s.a.C. § 346a.
[36] 15 UsC. §§ 2604-05.
[37]Alyssa Katzenelson & Scott Faber,On Cosmetics Prophylactic, U.S. Trails More than Than 40 Nations, Envtl. Working Grp. (Mar. 20, 2019), https://www.ewg.org/news-and-analysis/2019/03/cosmetics-safe-u.s.-trails-more-40-nations.
[38] Eur. Comm'n, Annex 2: List of Substances Prohibited in Cosmetic Products (last update: 24/04/2018), http://ec.europa.eu/growth/tools-databases/cosing/pdf/COSING_Annex%20II_v2.pdf (last accessed Mar. viii, 2019).
[39] Gov't of Canada,Cosmetic Ingredient Hotlist: Prohibited and Restricted Ingredients,
https://world wide web.canada.ca/en/wellness-canada/services/consumer-product-safety/cosmetics/cosmetic-ingredient-hotlist-prohibited-restricted-ingredients.html (terminal visited Mar. 2019); Eur. Comm'n, Annex 2: List of Substances Prohibited in Cosmetic Products (last update: 24/04/2018), http://ec.europa.eu/growth/tools-databases/cosing/pdf/COSING_Annex%20II_v2.pdf (last visited Mar. eight, 2019).
[40] Eur. Comm'northward, Annex 3: List of Substances Which Cosmetic Products Must Not Incorporate Except Bailiwick to the Restrictions Laid Down (last update: 24/10/2018), http://ec.europa.eu/growth/tools-databases/cosing/pdf/COSING_Annex%20III_v2.pdf (last accessed Mar. viii, 2019); Eur. Comm'n, Annex Five: List of Preservatives Allowed in Cosmetic Products (last update: 23/11/2018), http://ec.europa.european union/growth/tools-databases/cosing/pdf/COSING_Annex%20V_v2.pdf (concluding accessed Mar. 8, 2019).
[41]Eur. Chemicals Bureau, Annex XVII to Attain – Conditions of Restriction: Entry 68 Perfluorooctanoic Acrid (PFOA), https://world wide web.echa.europa.eu/documents/10162/7a04b630-e00a-a9c5-bc85-0de793f6643c (concluding accessed Mar. 8, 2019). The restriction comes into event in July 2020.
[42] In Oct 1951, the Business firm of Representatives authorized a Select Committee, led by then-Rep. James Delaney (D-N.Y.) to investigate the use of chemicals, compounds and synthetics in the production of cosmetics and related health furnishings.See H.R. Rep. No. 82-2182. More than a dozen bills to reform cosmetics accept been introduced since and so.See,e.g., the Cosmetics Prophylactic Human activity, S. 683, 93rd Cong. (1st Sess. 1973); H.R. 1527, 93rd Cong. (1st Sess. 1973) (requiring that cosmetics containing mercury or any of its compounds comport labeling stating that fact); H.R. 14805, 93rd Cong. (1st Sess. 1974) (authorizing FDA to halt the sales and distribution of food, drugs, and cosmetics adulterated or misbranded in a manner that presents an imminent take chances to the public health); H.R. 6249, 94th Cong. (1st Sess. 1975) (applying the provisions of the FDCA to hair dyes); the Cosmetics Safe Amendments of 1975, Southward. 1681, 94th Cong. (2nd Sess. 1976); Cosmetics Act, H.R. 1993, 95th Cong. (1st Sess. 1977); Cosmetics Safety Amendments, S. 2365, 95th Cong. (1st Sess. 1977); Nutrient, Drug, and Cosmetics Amendments of 1980, H.R. 2554, 91st Cong. (1st Sess. 1980) (permitting the inspection of a consulting laboratory in which food, drugs, devices, or cosmetics are beingness processed, packed, or held); the Rubber Cosmetics Human action of 2010, H.R. 5786, 111th Cong. (2d Sess. 2010); the Safe Cosmetics Act of 2011, H.R. 2359, 112th Cong. (1st Sess. 2011); Cosmetics Safety Enhancement Human activity of 2012, H.R. 4262, 112th Cong. (second Sess. 2012); Cosmetics Safety Amendments of 2012, H.R. 4395, 112th Cong. (2nd Sess. 2012); Safe Cosmetics and Personal Care Products Act of 2013, H.R. 1385, 113th Cong. (1st Sess. 2013); Personal Intendance Products Safety Human activity, S. 1014, 114th Cong. (1st Sess. 2015); Cosmetics Modernization Amendments of 2015, H.R. 4075, 114th Cong. (1st Sess. 2015); Cosmetics Modernization Amendments of 2017, Personal Care Products Condom Act, S. 1113, 115th Cong. (1st Sess. 2017); Personal Care Products Safety Act, S. 726, 116th Cong. (1st Sess. 2019).
[43] The following companies support bipartisan cosmetics reform legislation: Amyris (Biossance), Au Naturale, Babo Botanicals, Beautycounter, California Infant, Coalition of Handcrafted Entrepreneurs, Cote, Earth Mama Organics, Éclair Naturals, Estee Lauder Companies, EO Products, Goddess Garden Organics, Handcrafted Soap & Corrective Guild, Handmade Cosmetic Brotherhood, Herban Lifestyle, the Honest Company, Johnson & Johnson, Juice Beauty, 50'Oreal, Fabricated Of, Makes 3 Organics, Milk + Honey, MyChelle Dermaceuticals, OSEA, Peet Rivko, Procter & Take a chance, Rahua, Revlon, Seventh Generation, Silk Therapeutics, SkinOwl, S.Westward. Basics, Tenoverten, Unilever, Vapour Organic Dazzler.
[44] Mark Mellman & Linda DiVall,Findings From a National Survey of Likely 2016 General Election Voters (February. 2016), https://cdn.ewg.org/sites/default/files/u381/cosmetics.pdf?_ga=1.55566627.92668946.1470953450.
Source: https://www.ewg.org/the-toxic-twelve-chemicals-and-contaminants-in-cosmetics
Posted by: jonesorid1941.blogspot.com

0 Response to "What Is In Your Makeup? Lead Chemicals"
Post a Comment